What is TB and how is it transmitted?
Tuberculosis (TB) is a chronic, primarily respiratory infectious disease of mammals caused by a group of closely related bacteria known as the Mycobacterium tuberculosis (MTB) complex. This group includes:
- M. bovis – responsible for TB in cattle (bovine TB) and other mammals, including occasionally people. Cattle are the natural host of M. bovis, but nearly all warm-blooded animals, including goats, deer, pigs, camelids, sheep, cats and badgers are susceptible to the infection. This broad range of animal hosts complicates the eradication of bovine TB.
- M. tuberculosis – the primary cause of TB in people, although it rarely causes disease in animals.
This guidance is mainly concerned with TB caused by M. bovis and all further references to ‘TB’ denote the disease resulting from infection with M. bovis.

South American camelids (llamas, alpacas, vicuñas and guanacos) are susceptible to M. bovis and can develop lesions and clinical signs of TB. The infection can spread either directly (animal to animal) or indirectly via materials contaminated with the secretions or excretions of infectious animals. This can include contact with:
- infected cattle to other domestic animals (e.g. other camelids);
- wildlife reservoirs of infection; or
- contaminated, pasture, equipment, feedstuffs, water and slurry.
Infection with M. bovis is also a zoonosis, meaning that it can be naturally transmitted from vertebrate animals to humans. Camelids infected with M. bovis can also potentially transmit the bacterium to people who come into close contact with them and inhale the bacteria shed by infectious animals in respiratory and other secretions, or through contamination of unprotected cuts or abrasions in the skin while handling infected animals or their carcases. Even so, the risk of camelid-to-human transmission of M. bovis is considered to be low, with only two cases documented in Great Britain so far.
A small number of cases of TB are reported in camelid herds each year in Great Britain (GB), mainly in higher bTB incidence areas of England and Wales. Some of those TB incidents in alpaca herds have been quite severe, leading to complete or partial depopulation of the affected holdings. Official statistics relating to bTB in camelids (and other non-bovine livestock species) in GB are published approximately every quarter on GOV.UK.
Signs of TB in camelids
TB is difficult to diagnose in camelids on clinical examination alone. Camelids infected with M. bovis may not show any clinical signs, and if they do, those signs are common to many other diseases. Furthermore, the incubation period of the disease is highly variable and can range from a few months to many years, in which infected animals do not exhibit any obvious clinical signs. However, once the first clinical signs appear, the diseased animals tend to deteriorate quite rapidly.
TB should be considered in cases of chronic loss of condition and appetite, and debilitating disease (with or without respiratory signs). A chronic cough can be indicative of TB in camelids and should in particular be considered when a camelid has failed to respond to antibiotic treatment for a respiratory infection. Enlarged lymph nodes (glands) is another possible sign of TB.

In the first instance, camelid keepers concerned about the health of any animal on their premises should always seek advice from their private vet. Post mortem examination of all unexplained camelid deaths is also strongly recommended. If, following a clinical or post-mortem examination, the private vet suspects that an animal has TB, the law requires that they must notify the Animal and Plant Health Agency (APHA) without delay and that the diseased animal must not be treated with antimicrobial drugs for TB. APHA will then undertake an investigation and, if TB cannot be ruled out, restrictions may be imposed on the movement of animals from the premises pending the completion of diagnostic tests.
TB testing of camelids
Sensitivity and specificity explained
Diagnostic sensitivity and specificity are terms used to describe the accuracy of a test i.e. its ability to correctly identify infected and uninfected individuals as positive and negative, respectively.
Sensitivity is the probability that a test will correctly identify an infected animal as positive. The higher the sensitivity, the lower the probability of incorrectly classifying an infected animal as uninfected (a false negative result).
Specificity is the probability that a test will correctly identify an animal that is free from infection as negative. The higher the specificity, the lower the probability of incorrectly classifying an uninfected animal as infected (a false positive result).
Q&A
No. There is currently no statutory routine TB surveillance programme for camelids in GB. Whilst camelids are not routinely tested for TB, APHA can serve a notice on any camelid keeper requiring the animals to be tested by a certain date at the government’s expense to ascertain the presence of TB. This may be necessary, for instance, in one of the following circumstances:
- for ‘diagnostic’ purposes, e.g. when suspect lesions of TB have been reported during post-mortem examination of a dead camelid and/or M. bovis has been identified by polymerase chain reaction (PCR) testing or (if necessary) bacteriological culture from tissue samples, to check the TB status of other animals in the herd of origin;
- when infection with M. bovis has been detected in a cattle herd or other livestock adjoining (i.e. contiguous to), or co-located with a camelid herd;
- to allow removal of movement restrictions following the detection of TB test positive animals, TB clinical cases or TB post-mortem cases in a herd;
- when camelids have moved into a herd from another herd in which M. bovis infection has been confirmed (forward or spread tracings);
- to check the TB status of a camelid herd from which tuberculous animals have been moved to and detected in another herd (back or source tracings);
- one-off check testing of camelid herds in confirmed TB hotspots in the Low Risk Area of England.
A voluntary, private TB surveillance scheme endorsed by Defra and the camelid societies is also available in England, whereby camelid keepers can arrange for their animals to be privately tested for TB by their own vet at their own expense, for instance before or after animals are moved into a herd (pre-or post-movement testing). The Scottish and Welsh Governments also allow private testing of camelids for TB in Scotland and Wales, but this is not part of a TB surveillance scheme as for England. In GB, all private TB testing requires prior permission from or notification to APHA (depending on the type of test and country) and the results of such tests must be reported to APHA. Private testing is not appropriate if there is clinical suspicion of TB. If a camelid presents with clinical signs of TB then you must notify APHA immediately.
Although they are susceptible to M. bovis, the reported incidence of disease in camelids in GB is low and they are considered incidental ‘spillover’ hosts of M. bovis. This means that TB occurs within the species only as long as there is input from an external source (maintenance host), in this case cattle and/or badgers. In most cases camelids become infected through direct or indirect contact with maintenance hosts. There have been instances of translocation of TB through the movement of undetected M. bovis-infected animals between camelid herds. Therefore, camelids can potentially behave as amplifiers (vectors) of infection for other animals. However, generally speaking, camelids pose a low risk of transmitting M. bovis infection to cattle and badgers, and as such they do not have a significant role in the epidemiology of bTB in GB. There is also no statutory requirement to officially identify camelids or record their movements. The costly routine surveillance TB testing carried out for cattle would be disproportionate for camelids, and the current lack of mandatory registration and identification requirements would make such a programme difficult to manage and enforce.
The tuberculin skin test is used worldwide for TB screening of camelids. The skin test in camelids involves injecting a small amount of ‘bovine’ tuberculin (a sterile, purified mixture of proteins extracted from cultures of M. bovis) into the deep layer of the skin of the chest wall behind the elbow. The skin is first clipped and the thickness of the skin fold measured in millimetres using special callipers before the tuberculin is injected. After 72 + 4 hours, the vet returns and re-measures the skin at the site of the injection to determine if the animal is classified as a reactor or negative (pass). Avian tuberculin may be injected alongside bovine tuberculin as a comparative test to provide a readout with higher specificity, for instance in situations where it has not been possible to confirm M. bovis infection.
Studies have been conducted to evaluate the performance of the comparative tuberculin skin test in camelids. A protocol for the skin test in camelids was published in 2021 by the European Reference Laboratory for bTB for use by EU member states. However, APHA’s experience in GB is that the tuberculin skin test is a relatively poor indicator of TB infection in camelids due to its very low sensitivity in these species (estimates are as low as 5-15% and probably 25% at best1).
That is why TB antibody blood tests have been adapted, optimised and validated at APHA to supplement the tuberculin skin test in these species and improve the detection of M. bovis-infected alpacas and llamas. These antibody tests are carried out 10-30 days after the injection of tuberculin in animals that have not reacted to the tuberculin skin test. Nonetheless, the tuberculin skin test still has an important role in boosting the sensitivity of TB antibody tests in camelids (see below). TB diagnostic tests in GB are used in line with recommendations in the TB chapter of the Manual of Diagnostic Tests and Vaccines for Terrestrial Animals as adopted by the World Organisation for Animal Health.
In GB three antibody blood tests have been validated and officially approved for statutory and private TB screening of live camelids. In Wales only, the Enferplex test is not approved for statutory testing, but can be used for private testing. These tests can in turn be combined (and their results interpreted) in different ways depending on the prior likelihood of M. bovis infection being present in the herd or animal being tested.
Antibody blood tests for use in camelids
The Dual Path Platform (DPP) VetTB test (Chembio Diagnostic Systems, Inc. USA) is based on the detection of antibodies using a set of recombinant (artificially manufactured) M. bovis antigens, MPB83 and ESAT6/CFP10, on two separate antigen lines of a lateral flow cassette. Its use was first validated in an APHA study supported by the British camelid industry and published in a peer-reviewed scientific journal in 2012. Test performance was subsequently re-evaluated and updated by APHA in 2018.
The IDEXX ELISA test for bTB (IDEXX Laboratories Inc., USA) involves the detection of specific antibodies using a cocktail of M. bovis antigens (MPB83 and MPB70), and a simple ELISA (enzyme-linked immunosorbent assay) platform. The IDEXX ELISA was added to the World Organisation for Animal Health (OIE) registry of tests for bTB in 2012. APHA adapted this bovine test for camelids and its use was validated in an APHA study supported by the camelid industry and published in a peer-reviewed scientific journal. Test performance was subsequently re-evaluated and updated by APHA in 2018.
The Enferplex (multiplex) ELISA (Enfer Scientific) is based on the detection of antibodies against seven M. bovis antigens (MPB70, MPB70peptide, PPDB, ESAT6, CFP10, MPB83, Rv3616c) spotted separately on an ELISA platform. Its use was first validated in an APHA study supported by the British camelid industry and published in a peer-reviewed scientific journal in 2012. Test performance was subsequently re-evaluated and updated by APHA in 2018.
The following tables present the diagnostic sensitivity and specificity of these tests deployed on their own, or in dual-test combinations. These data are taken from APHA re-evaluation of DPP, IDEXX & Enferplex test (S Rhodes’ study report – March 2018) (abridged).
Table 1 – Sensitivity and specificity of the individual tests
| Antibody test | Sensitivity [95%CI] | Specificity [95%CI] |
|---|---|---|
| DPPVetTB | 56% [46.3-65.7] | 98.3% [96.8-99.8] |
| IDEXX | 74% [65.4-82.6] | 97.7% [95.3-99.1] |
| Enferplex 2-spot | 67% [57.8-76.2] | 99.3% [98.4-100] |
Statistical comparison showed the DPP VetTB test to have a significantly lower sensitivity when compared to the IDEXX test, but not when compared to the Enferplex 2-spot test. There was no statistical difference in sensitivity between the IDEXX and Enferplex-2-spot tests. There was no significant difference in test specificities of the individual tests.
The private Enferplex test is currently not offered by APHA and must be organised with SureFarm Ltd.
Table 2 – Sensitivity and specificity of parallel / high sensitivity test options
| Antibody test option | Sensitivity [95%CI] | Specificity [95%CI] |
|---|---|---|
| IDEXX & DPPVetTB | 74% [65.4-82.6] | 96% [93.8-98.2] |
| Enferplex 2-spot & DPP VetTB | 71% [62.1-79.9] | 96.7% [95.8-99.4] |
| IDEXX & Enferplex 2-spot | 75% [66.5-83.5] | 97.3% [95.4-99.1] |
For the combined antibody tests with parallel interpretation, a positive readout for either of two tests provides a positive combined parallel test result. Statistical comparison showed no significant difference in sensitivity or specificity between any of the parallel test combinations.
Table 3 – Sensitivity and specificity of serial / high specificity test options
| Antibody test option | Sensitivity [95%CI] | Specificity [95%CI] |
|---|---|---|
| Enferplex 4-spot | 60% [50.4-69.9] | 99.66% [98.9-100] |
| IDEXX & DPPVetTB | 56% [46.3-65.7] | 100% |
For the combined IDEXX and DPP VetTB serial interpretation test, both tests must be positive to generate a positive combined serial test result. Statistical comparison showed no significant difference in test sensitivity or specificity between the Enferplex-4-spot test and the serial combined IDEXX and DPP VetTB test.
Q&A
All three blood tests (DPPVetTB, IDEXX and Enferplex) are based on the detection of serum antibodies to a set of recombinant (artificially manufactured) M. bovis antigens. Antigens are substances produced by bacteria and other pathogens that stimulate the host’s immune system to produce antibodies (proteins) against them.
When an animal is infected with M. bovis, its immune system produces antibodies against immunogenic components (antigens). These can include antibodies against both surface and/or secreted antigens of the bacterium. The three blood tests expose the animal’s blood (serum) sample to specific synthetic M. bovis antigens, which generally represent the most commonly recognised antigens, to see if antibodies specific to these antigens are present in the serum.
If the animal has been infected with M. bovis and developed an antibody response against the bacterium, then the antibodies present in the blood sample taken from the animal will bind to the synthetic M. bovis antigens used in the test kit and this antibody-antigen complex will be detected/visualised through a chemical reaction. By contrast, if the animal has not been infected with M. bovis then the sample will not contain specific antibodies that can bind to the M. bovis antigens in the test kit.
Yes. The diagnostic accuracies of the three approved antibody tests for TB in camelids (DPPVetTB, IDEXX and Enferplex) were first estimated as part of a field trial conducted with the support of the British camelid societies in 2010-2011. The results of this validation trial were peer-reviewed and published in 2012. The performance characteristics of these tests under GB conditions were re-evaluated by APHA in March 2018.
Yes, in England and Scotland. In Wales, the Enferplex test can only be used for private testing and is not approved for statutory testing. The diagnostic sensitivities and specificities of the three antibody tests for TB in camelids are comparable and not statistically different. Therefore, when carrying out voluntary or statutory TB testing of camelids in England and Scotland, keepers can choose which antibody tests to use, depending on whether the objective is to maximise detection of infected animals (for PCR or culture positive herds under TB restrictions) or minimise the probability of false positive results (for contiguous testing of unrestricted herds). In Wales only, the use of the Enferplex test as an alternative to the DPPVetTB and IDEXX tests can only be approved for private non-statutory testing. For statutory testing by APHA, the DPPVetTB and IDEXX test are used, either in serial or parallel interpretation.
In England and Scotland, there are certain restrictions on test availability regarding the number of samples for each blood test option. The DPP VetTB test is reserved for submissions of 40 or fewer samples. Larger sample submissions are tested using the IDEXX/Enferplex ELISA test combinations. An Enferplex test run requires 10 or more samples to be cost-effective. The laboratory will inform the camelid keeper if this is likely to cause a delay in test reporting, and may offer an alternate, equivalent test option if this is the case.
The high specificity test options require both tests to be positive to generate a positive combined serial test result. High specificity test options are used in the following scenarios where it is desirable to minimise false positive results:
- Private voluntary TB blood testing of camelids, e.g. domestic and international animal movements, purchases, shows, matings.
- Where bTB is strongly suspected in a camelid herd (e.g. characteristic TB lesions detected in one or more animals at post mortem examination) pending identification of M. bovis in the laboratory.
- Camelid herds sharing a holding (co-located) with a cattle TB breakdown herd with lesion and/or PCR/culture-positive animals or with other farmed non-bovine animals affected by a PCR/culture-positive TB incident.
- Camelid herds not under movement restrictions, but contiguous (i.e. share a common land border) to a holding with a cattle TB breakdown herd with lesion and/or PCR/culture-positive animals, or to non-bovine farmed animals affected by a PCR/culture-positive bTB incident.
- Camelid herds not under movement restrictions but undergoing TB testing due to an epidemiological link with a known infected herd e.g. back (source) tracing.
- Official re-testing by APHA of camelids with two consecutive positive results on a private Enferplex test read at the 2-spot interpretation.
Note that in Wales, APHA only uses the DPPVetTB/IDEXX test combination only for official TB testing of camelids in serial interpretation. This includes the official re-testing of 2-spot positives from a private Enferplex test.
The high sensitivity test options require either of the two tests to be positive to generate a positive combined parallel test result. High sensitivity test options are used in scenarios where M. bovis infection has been confirmed by PCR testing or (if necessary) bacteriological culture. It is desirable in these circumstances to maximise identification of TB-infected camelids and minimise false negative results. High sensitivity test options are used in the following scenarios:
- TB breakdown situations where bovis infection has been confirmed in the herd by PCR testing or bacteriological culture. In Wales, this also includes camelid herds considered high risk on epidemiological grounds but is dependent on test results.
- Forward (spread) tracing and blood testing of individual animals from a camelid herd where bovis infection has been detected by PCR testing or bacteriological culture.
Note that the Enferplex test option is not available for statutory parallel testing of camelids located in Wales.
The purpose of a tuberculin skin test carried out before blood testing is to boost the levels of specific antibodies generated by the animal’s immune system against a pre-existing M. bovis infection. The injection of bovine tuberculin essentially amplifies the level of antibodies that already exist as a result of M. bovis infection and this amplification increases the sensitivity of the test. These antibodies might otherwise be below the detection threshold if a skin test is not carried out, thereby producing a false-negative test result. For more information see APHA’s paper on the anamnestic boosting effect of the skin test on antibody responses to M. bovis in camelids – summary of the evidence. For the sole purpose of boosting prior to a blood test, a single intradermal injection of bovine tuberculin need only be carried out and the result of the test does not need to be read or recorded. In some scenarios a comparative skin test is recommended or required before blood testing, so keepers are advised to check with their private vet and/or APHA. Further information about the different statutory TB testing scenarios for camelids in England and Scotland is presented in the flow diagrams above. In Wales, for statutory TB testing of camelids, a skin test is always carried out and read prior to an antibody test.
No. The injection of tuberculins (bovine or avian) does not induce a false positive antibody response in animals that are not infected with M. bovis. The tuberculin skin test involves the injection into an animal’s skin of a very small dose (0.1ml) of a purified, ‘cocktail’ of proteins extracted from heat-killed liquid cultures of bovine TB bacteria. As mentioned above, its main purpose in serological testing is to boost the levels of pre-existing specific antibodies generated by M. bovis infection, which might otherwise be below the detection threshold thereby producing a false-negative test result. The injection of tuberculin essentially amplifies the antibodies already induced by an existing infection. It does not trigger the production of a new antibody response in TB-free animals as there are no pre-existing antibodies to boost. Tuberculin does not build up in an animal from one skin test to the next.
Camelid TB testing scenarios
The following set of flow diagrams summarise the process and actions taken by APHA for statutory and voluntary TB testing scenarios in camelids in England and Scotland.
These scenarios do not cover every situation, and veterinary judgement can overrule the below guidance depending on the case.
Post-mortem examinations are carried out to assess the stage and severity of the infection at the time of slaughter and, where required, to obtain tissue samples for PCR test, culture and DNA typing of M. bovis to assist with epidemiological investigations. However, TB can only be detected directly by PME if the infection has caused lesions in the infected organ(s) and associated lymph nodes that are sufficiently extensive to be visible to the naked eye.
There are three possible outcomes from a PME on a TB test-positive or suspect animal:
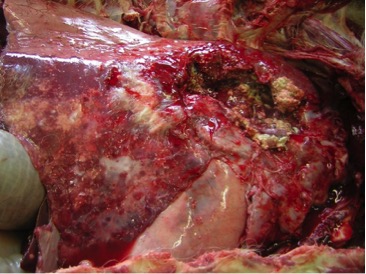
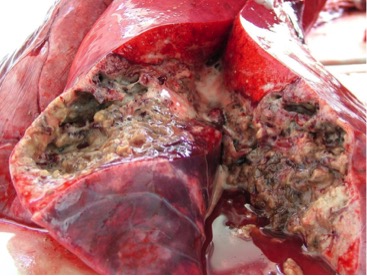
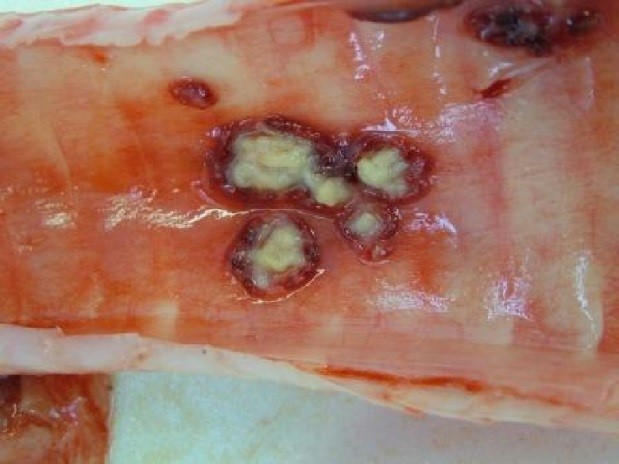
- Typical visible lesions: tissue changes characteristic of M. bovis infection (with common colours and consistency) were seen.
In South American Camelids, a lesion is reported as typical only if it was detected in organs of the respiratory tract (lung and associated lymph nodes).
- Atypical visible lesions: tissue changes seen cannot be definitively attributed to M. bovis infection, but the involvement of the bacterium cannot be ruled out either.
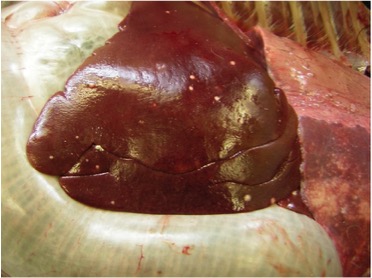
In South American Camelids only, the recording of atypical visible lesions indicates that TB-like tissue changes were detected at other anatomical sites, e.g. liver, head or intestinal lymph nodes, in the absence of visible lesions in the lungs and/or chest lymph nodes. Such lesions are subject to additional microscopical examination. If organisms with characteristic mycobacterial morphology are identified, then the lesions are reported as visible and typical.
- No visible lesions: no tissue changes visible to the naked eye could be detected.
Please note that TB skin and antibody blood tests are more sensitive than PME and may detect the immune response to the bovine TB bacterium before the development of any visible lesions and clinical signs of TB. In some cases, the microscopic lesions of TB in the affected organ(s) can also take a very long time to progress to larger, visible lesions, if at all. Therefore, failure to detect TB-like lesions at PME does not mean the animal was free of infection.
* PCR test results are usually reported to the camelid keeper within three weeks. In the rare instances where a valid PCR test result is not obtained, microbiological culture of M. bovis will be attempted and this can take up to 22 weeks for a final result. In the meantime, a comparative skin test of the herd may be carried out.
In exceptional cases, where there is strong evidence of infection, a check blood test may also be carried out pending final culture results to avoid delay that may exacerbate the problem.
The owner can choose:
- Enferplex (4-spot with interpretation using statistical package) provided by SureFarm Ltd.
OR
- Combined DPPVetTB & IDEXX (using serial interpretation) provided by APHA
In cases where there are subsequent positive PCR/culture results, the results of the skin or blood tests may be re-interpreted and further animals removed to better manage the risk of further spread of infection within the herd.
Please note that the skin and blood check tests are not qualifying tests for the purposes of withdrawing herd movement restrictions as the skin test would have been performed less than 90 days after the death/removal of the index case on the holding. Any animal which is positive to a skin or blood check test must be isolated and removed. If the subsequent PCR/culture result is negative (i.e. full parallel testing is not triggered), the herd would be subject to a comparative skin test at least 90 days after the removal of the last test reactor. If that follow up skin test is negative, APHA will lift movement restrictions.
Once M. bovis infection has been confirmed in a camelid herd, APHA may not carry out post mortem examinations and PCR/cultures of every test-positive animal, particularly where there are large numbers of positives and post mortem results are not essential to establish the next step in managing the TB incident.
** After the initial round of government-funded parallel TB antibody blood testing in camelid herds affected by M. bovis-culture positive breakdowns, APHA may consider, on a case-by-case basis, requests from herd keepers for additional, privately funded TB Enferplex blood tests of skin and DPP/IDEXX test-negative animals if the herd in question does not immediately qualify for a further round of antibody blood testing conducted by APHA. All TB seropositive animals in such herds will be removed with compensation, whether the blood tests were funded by APHA or the herd keeper.
*For contiguous camelid herds, a single intradermal injection of bovine tuberculin needs to be administered 10 to 30 days prior to the blood test. Its only purpose is to boost the specific antibody response to M. bovis in any TB-infected camelids in the herd, thus increasing the sensitivity of the ensuing blood test. If the camelid owner wishes, APHA may instruct the private vet to carry out a comparative skin test instead of the intradermal injection of bovine tuberculin for boosting only, but the skin test reactions to the avian and bovine tuberculins must be read, recorded on the Tuberculin Test Chart for Non-Bovine Animals (TN52A) and Non-Bovine – Tuberculin Test Report and Certificate of Clinical Inspection (TN52B) and acted upon if an animal is deemed a reactor.
For co-located camelid herds and back-traced camelids, a comparative skin test must be carried out.
* A positive Enferplex test result is defined as a positive result to the 4-antigen test. For the combined IDEXX and DPP VetTB serial interpretation test, both tests must be positive to generate a positive combined serial test result. No inconclusive results are recorded for the combined IDEXX and DPP VetTB test. Any camelid which is positive based on the 2-antigen Enferplex test, but negative based on the 4-antigen test is regarded as inconclusive. The exception to this is where the proportion of 2-antigen results exceeds the threshold set by the SureFarm statistical package, in which case SureFarm will inform APHA who will decide on appropriate actions on the test positive animals, based on the herd’s location and TB testing history. When retesting inconclusive animals, if a skin test (comparative or bovine only) was carried out 10-30 days before the original blood sampling and a subsequent comparative skin test is required by APHA as part of retesting, this must be carried out at least 90 days after the first skin test.
** Voluntary antibody testing is recommended annually for at least the first two years. Thereafter, herds in low incidence areas may choose to revert to four-yearly testing after two or more rounds of annual herd tests with negative results, unless animal movements from camelid herds outside the health scheme occur.
*** PCR test results are usually reported to the camelid keeper within three weeks. In the rare instances where a valid PCR test result is not obtained, microbiological culture of M. bovis will be attempted and this can take up to 22 weeks for a final result.
Export
For guidance on the export of South American camelids to the EU or Northern Ireland, please see the government’s current export health certificate (EHC) and associated Notes for Guidance. For exports to third countries, see the EHC search tool on gov.uk.
Post mortem examination
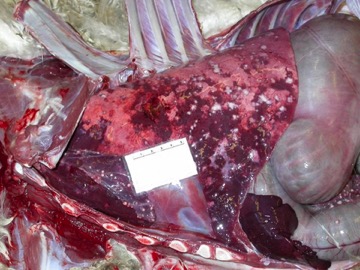
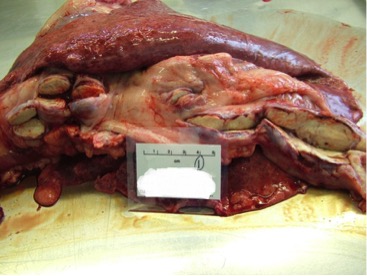
Post mortem images of TB in camelids
At post mortem examination, if present, lesions characteristic of TB are predominantly found in the respiratory system although they are seen in other organs in cases of more generalised disease. Lung lesions are often very extensive and cavitation is common, where lung tissue is destroyed and replaced by a cavity.
Q&A
Cattle and other livestock that test positive for TB are normally removed to a slaughterhouse where their carcases undergo post-mortem meat inspection by the Food Standards Agency meat inspection team, who may also collect suitable samples from the carcase for bacteriological culture in an APHA laboratory (if required). Camelids, by contrast, are usually euthanased on farm and their carcases transported to an APHA laboratory for post-mortem examination (PME) and collection of tissue samples for PCR testing and (if necessary) bacteriological culture. Not every TB test positive camelid is subject to a PME and PCR testing and/or bacteriological culture, for instance in herds with multiple TB positive animals in which M. bovis has already been isolated during an ongoing TB incident. APHA veterinary pathologists follow a Standard Operating Procedure that covers all the relevant organs and lymph nodes to examine for TB lesions. As a result of that PME, the carcase is classified as having either:
- typical visible lesions (VL) usually in head and respiratory tract tissues and associated lymph nodes, or
- atypical lesions (AL) denoting TB-like lesions outside of respiratory tract organs and its associated lymph nodes; or
- no visible lesions (NVL).
If there are visible or atypical lesions in the carcase, then samples of the lesioned tissue are selected for PCR testing and (if necessary) bacteriological culture to identify the strain of M. bovis. If no visible lesions are found, then a mixed sample of lymph nodes taken from the thoracic and head lymph nodes (“NVL pool”) are submitted instead. More information about the PME process can be found on APHA’s science blog.
Lesions in a carcase can present as TB lesions to the naked eye but cannot be confirmed as TB until PCR testing and/or bacteriological culture (and genetic typing of any isolates) has been performed. For camelids, when lesions characteristic of TB are only found in anatomical sites outside the respiratory tract, they are classified as atypical. This does not mean that the lesions were not caused by M. bovis as there is no way of distinguishing them by their gross appearance alone.
For high specificity antibody tests the lesion rate is about 74% of test-positives and for high sensitivity tests, it’s about 48% of test-positives. See APHA post mortem examination information for TB seropositive camelids compulsorily slaughtered from 2014 to 2020.
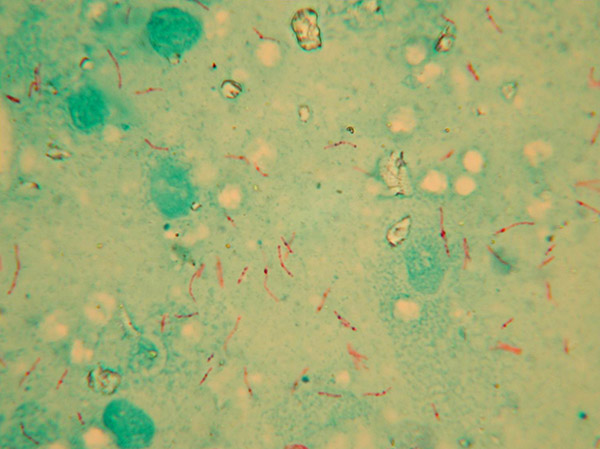
On 30 March 2022, APHA adopted a new polymerase chain reaction (PCR) test for direct detection of M. bovis in tissue samples collected at post-mortem examination. The PCR test has replaced bacteriological culture as the primary confirmatory test of M. bovis infection in camelids at APHA laboratories. However, the target DNA sequence amplified through PCR testing is not suitable to perform whole genome sequencing (genetic typing) to identify the strain of the bacterium. Therefore, bacteriological culture is still carried out by APHA in selected PCR-positive animals that satisfy certain criteria.
Whenever bacteriological culture is required, a small sample of tissue collected at post-mortem examination is processed and incubated in solid and liquid media to encourage the growth of M. bovis in the laboratory, if the bacterium is present in the sample. Bacteriological confirmation of M. bovis infection is highly dependent on the presence of any visible lesions of TB in the carcase. In cattle, around 95% of VL tissue samples will grow M. bovis. By contrast, when a pool of lymph nodes without visible lesions of TB is harvested, the probability of recovering M. bovis drops to around 5%. If M. bovis is cultured, whole genome sequencing (genetic typing) is carried out to identify the strain of the bacterium.
In a new TB breakdown, samples collected from post-mortem tissues are PCR tested and (if necessary) subjected to bacteriological culture to identify the particular strain of M. bovis where possible, to aid infection tracing. Subsequent test-positive camelids removed during the same ongoing breakdown may not be PCR tested and/or cultured as this information has already been collected.
No. It’s not possible to conclude that any animal that returned a positive serology test result to a validated TB test was not infected. Research data tells us the likelihood is that they were infected regardless of the results of the PME results. As displayed in the table above, for high specificity blood tests around 25% of test-positive camelids were NVL, while for high sensitivity tests around 52% were NVL. However, NVL at PME, particularly after a positive test result does, not mean absence of M. bovis infection. The post mortem diagnostic methods for TB are less sensitive than the immunological (particularly antibody) tests that vets use to detect the infection in live animals. The sensitivity of post mortem examination for identifying TB lesions depends upon several factors, including the bacterial load in the carcase of the test-positive animal and the progression of infection to the point where lesions are detectable by the naked eye. Progression of TB can take weeks to many years, whereas the ante-mortem skin and blood tests can detect an immune response to the bacterium after a matter of weeks or months. While a positive PCR test and/or culture result for M. bovis will confirm that the animal was infected, NVL at post mortem and negative PCR test and/or culture results, particularly after a positive ante-mortem test, does not rule out M. bovis infection or negate the positive test results.
Compensation

Compensation rates for camelids compulsorily slaughtered for bTB control in GB are determined under the relevant domestic legislation for each country:
- In England, the Tuberculosis (Non-bovine animals) Slaughter and Compensation (England) Order 2017
- In Wales, the Tuberculosis (Wales) Order 2011
- In Scotland, the Tuberculosis in Specified Animals (Scotland) Order 2015
For camelids compulsorily slaughtered in GB, the rates of compensation paid are as follows:
- £750 for an animal 18 months old or younger
- £750 for a non-breeding animal over 18 months old
- £1500 for a stud male or breeding female over 18 months old

Free biosecurity advice for camelid keepers in England

The TB Advisory Service (TBAS) provides free, bespoke advice about practical, cost-effective measures to build herd resilience to bTB through on-farm advice visits and telephone advice. Keepers of farmed non-bovine animals across England are eligible for free advice from TBAS.
Camelid keepers can request a free one-to-one visit with an experienced veterinary advisor, who will provide bespoke recommendations to reduce the risk of bTB infection for herds that are currently TB-free, whilst also discussing measures to prevent repeated re-infection for herds experiencing a TB incident.
Contact TBAS via email at info@tbas.org.uk or over the phone on 01306 779410 to book your free advice visit.
Reference & resources
- Rhodes et al., 2015. Chapter 12; Mycobacterial infections in camelids. In: CABI publication ISBN-13: 978 1 78064 396 0
Tuberculosis, Leprosy and Mycobacterial Diseases of Man and Animals – The Many Hosts of Mycobacteria

Useful resources
- Information on how to deal with TB in non-bovine animals is available on GOV.UK
- British Alpaca Society (BAS)
- British Llama Society (BLS)
- Research Gate: Stevens JB, Theon CO, Rohonczy E, Tessaro S, Kelly HA, Duncan JR. 1998. The immunological response of llamas (lama glama) following experimental infection with Mycobacterium bovis.